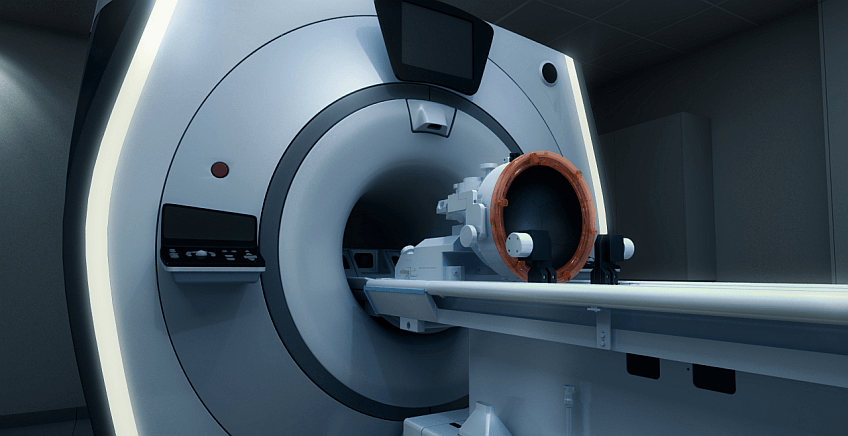
Medical device company Insightec has announced the creation of a new spin-out, Lotus Neuro, dedicated to developing advanced therapies for brain tumors. The move is based on Insightec’s focused ultrasound technology and its clinically demonstrated ability to temporarily and precisely open the blood–brain barrier (BBB), enabling more effective delivery of drugs to brain tissue. The new company marks a strategic deepening of Insightec’s brain-therapy activities and aims to translate clinical insights accumulated over recent years into targeted therapeutic applications. Lotus Neuro will operate in the field of neuro-oncology, with an initial focus on glioblastoma—one of the most aggressive and lethal forms of brain cancer.
Internal Leadership and a Familiar Investor: The Spin-Out Structure
Lotus Neuro will be led by Dr. Arjun J.J. Desai, currently Chief Strategy Officer at Insightec, who will assume the role of founder and CEO of the new company. His designation as founder suggests that the spin-out was driven from within Insightec itself. The company has not disclosed whether Lotus Neuro will be based in Israel or how many employees it will initially employ.
The venture will be funded by Nexus NeuroTech Ventures, an investment firm well known to Insightec’s management and to the market. Nexus was among the key investors that backed Insightec in 2024, during a period of severe financial challenges that included a “going concern” warning and liquidity constraints that threatened the company’s continued operations. At the time, Nexus participated in a capital raise of approximately $150 million, helping stabilize Insightec’s financial position and ensure business continuity.
While the announcement did not disclose the size of Nexus’s investment in Lotus Neuro or its ownership stake, Nexus is the only investor named at this stage—suggesting it may hold a significant share in the new company. The spin-out may also reflect a strategic move by Nexus to unlock additional value from its investment in Insightec by opening a new therapeutic market built on the same underlying technology.
Founded in 1999 and headquartered in Tirat Carmel, Israel, Insightec operates in the U.S. and Europe as well. The company developed the Exablate system, an MRI-guided focused ultrasound device used for non-invasive treatments of neurological conditions such as essential tremor and Parkinson’s disease, as well as certain brain tumors.
From Liquid Biopsy to Therapy: The Clinical Foundation of Lotus Neuro
The scientific roots of the spin-out lie in the LIBERATE clinical trial launched in 2022, in which Insightec explored the use of low-intensity focused ultrasound in glioblastoma patients to enable liquid biopsy. Brain tumors present a unique challenge due to the blood–brain barrier, a natural defense mechanism that complicates both diagnosis and effective drug delivery.
In the trial, researchers demonstrated for the first time that ultrasound activation could enable the release of tumor-derived genetic material into the bloodstream, allowing diagnosis and disease monitoring without invasive procedures. This capability—initially observed as a byproduct of a diagnostic study—became the foundation for the spin-out.
If ultrasound can influence the tumor environment to allow materials to move outward, the same mechanism can potentially be harnessed to deliver drugs and biological therapies more effectively into the tumor itself. This is where Lotus Neuro enters the picture. The company will focus on developing combinations of ultrasound with existing and novel drugs, aiming to enhance their effectiveness in brain cancers where treatment options remain severely limited.
Glioblastoma is widely regarded as one of the greatest challenges in modern medicine, with poor survival rates and few effective therapies. Any technology that improves access to the tumor or boosts the efficacy of existing drugs could represent a meaningful breakthrough. For Insightec, the spin-out allows the company to remain focused on its role as a platform and medical device provider, while Lotus Neuro operates as a therapy-development company with broader scientific, strategic, and commercial flexibility.
In the background stands Nexus, which recently raised a large fund worth hundreds of millions of dollars for investments in advanced technologies, positioning Lotus Neuro as part of a broader strategy focused on deep, long-term innovation. The spin-out reflects not only technological progress, but also a calculated business move to extract new value from a technology that has already proven itself in clinical settings.